Tensión de superficie. Peso y volumen de una gota de líquido
Esta calculadora encuentra el peso y volumen de una gota dada la tensión de la superficie, diámetro del tubo capilar y densidad del líquido.
Este contenido está bajo licencia de Creative Commons Attribution/Share-Alike License 3.0 (Unported). Esto significa que puedes redistribuirlo o modificar su contenido en forma libre bajo las mismas condiciones de licencia y debes mantener la atribución del mismo al autor original de este trabajo colocando un hipervínculo en tu sitio web a este trabajo https://es.planetcalc.com/2296/. Así mismo, por favor no modifiques o alteres ninguna de las referencias al trabajo original (si hubiera alguna) que se encuentre en este contenido.
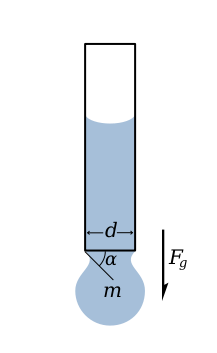
La imagen a la izquierda muestra una gota de líquido suspendida al final de un tubo por una tensión de superficie. Esta fuerza es proporcional a la longitud de la barrera entre el líquido y el tubo y es dada por
,
donde - coeficiente de tensión de la superficie en newtons por metro
La gota sigue suspendida siempre y cuando el componente vertical de la fuerza de superficie de tensión sea igual a la fuerza de gravedad, es decir
La máxima fuerza de tensión de superficie aplicada, cuando alfa es igual a 90º da el máximo peso de una gota de líquido con una tensión de superficie dada.
, entonces
Al tener la masa y densidad del líquido, el volumen es trivial.
En la realidad la gota se separa en donde el "cuello" se forma, justo debajo del final del tubo, entonces el diámetro utilizado debe ser menor al diámetro del tubo. Pero, si no se requiere alta precisión, entonces se pasa por alto.
También se estima en la calculadora el máximo diámetro del tubo de forma que permita que se formen gotas, la fuerza de tensión de superficie debe ser capaz de sostener al menos un hemisferio. Entonces dada la densidad del líquido, la forma termina siendo
y el radio es,
Un par de cosas sobre la calculadora; primero, no calcula el peso y volumen si el diámetro del tubo es mayor al dado por la ecuación anterior. Segundo, los valores por defecto de la densidad del líquido y coeficiente de tensión de superficie son del agua.
Comentarios